PHOSPHORUS-CONTAINING FLAME RETARDANTS
1. Overview
Phosphorus-containing chemicals are used as flame retardants for thermoset plastics, thermoplastics, paper, fibers, and paints. These include compounds such as red phosphorus, ammonium polyphosphate, organophosphate, phosphonate, phosphine oxide, chloroaliphatic and bromoaromatic phosphate. Their fire protection mechanism includes the formation of an insulating porous coal layer that protects the substrate from the effects of heat and flame and prevents the ingress of atmospheric oxygen, which interferes with the maintenance of the flame.
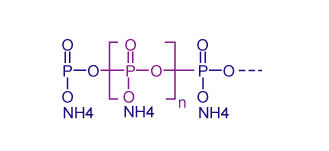
Photo 1: Structural formula of ammonium polyphosphate
Phosphorus has attracted interest as a flame retardant used to replace halogen-containing flame retardants because they are non-toxic and environmentally friendly. Although they are highly effective against fire in some cases, some resins such as polyolefine and styrene they do not appear to be effective against fire. Using synthetic phosphorus-bromide can reduce the halogen content used in polymers,
2. Fire retardant mechanism
The flame retardant mechanism of phosphorus depends on the type of phosphor compound and the structure of the polymer. They operate in either a condensed or a gas phase and can be a combination of both. Some phosphorus compounds will thermally decompose in the condensed phase to produce phosphoric and polyphosphoric acid. They can form a char on the surface that protects the polymer surface from flame and oxygen. In addition, they can dehydrate and ester polymers to form a considerable amount of porous coal. Phosphorus is known to promote the formation of porous coal. The layers of porous coal will prevent the contact of heat, flame, and oxygen to the substrate surface. The formation of inert gas during the construction of porous coal also plays an essential role in the fire resistance mechanism.
The combination of polyol, dipentearythriol, and phosphate compound together with a polymer will form a thick porous coal layer. Adding a gas-generating agent will form an expanded coal layer which protects the substrate material from heat, flame, and oxygen. As an example, the combination of ammonium polyphosphate, pentaerythritol, and melamine would form a flame retardant swelling coating.
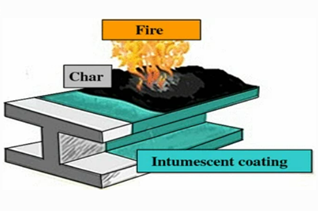
Photo 2: The mechanism of action of the anti-flaming paint
Organic phosphate ester is heat stable, high boiling point (>350oC), it is used as a plasticizer for cellulose acetate and poly(vinyl chloride), they are non-flammable and act as anti-plasticizer on fire. For example, poly(vinyl chloride) contains the plasticizer phthalate dioctyl which can burn in the presence of a flammable agent. In the replacement of organic plasticizers, organic phosphate ester will act as a flame retardant material.
Reaction (1) decomposition of phosphoric acid.
H3PO4 --> HPO2+HPO+PO (1)
H+PO --> HPO (2)
OH+PO --> HPO + H20 (3)
OH+ H2+PO --> HPO+ H2O (4)
The flame inhibition reaction is similar to the gas phase mechanism of the halogen compound shown in reaction (2). In reactions (3) and (4) they operate by the condensate phase mechanism, PO free radical reactions are especially important because it prevents the reaction from forming H+O2 --> OH+O.
Combustion can be inhibited by physical agents. If the concentration of inert gas from the flame retardant is high enough it acts as an inhibitor of flame formation.
Here's some basic knowledge. “FLAME RETARDANT CONTAINING PHOSPHOR” that we share. Hopefully, we will provide useful information to you. For more details, please contact Mega Vietnam New Materials Research and Development Center to have your questions answered and provide timely solutions.
>> Nhận tư vấn chuyển giao công nghệ sản xuất sơn chống cháy <<
Mọi thông tin vui lòng liên hệ:
Văn phòng: Tầng 2, A2-IA20, KĐT Nam Thăng Long, đường Phạm Văn Đồng, P. Đông Ngạc, Q. Bắc Từ Liêm, TP. Hà Nội, Việt Nam
Trung tâm nghiên cứu: Thôn Nhuế - xã Kim Chung - huyện Đông Anh - Tp. Hà Nội - Việt Nam.
Website: megaradcenter.com
Tel/Fax: (+84) 24 375 89089 / (+84) 24 375 89098
Email: contact@megavietnam.vn




